Tiny pieces of magnesium and fresh calcium are given to students to react in a test tube with a small amount of hydrochloric acid. Because calcium is slightly more reactive in acid than the magnesium, a visually more extreme reaction should occur.
Equipment required (per set):
- Hydrochloric acid (1 mol dm¯³) (Irritant)
- Small pieces of magnesium ribbon (highly flammable)
- Small pieces of fresh calcium (highly flammable)
- 2 Test tubes
- Test tube rack
- Splint
The equations for each reaction are below:
Magnesium + hydrochloric acid 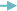 magnesium chloride + hydrogen
magnesium chloride + hydrogen
Mg + 2HCl  MgCl2 + H2
MgCl2 + H2
Calcium + hydrochloric acid 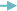 calcium chloride + hydrogen
calcium chloride + hydrogen
Ca + 2HCl  CaCl2 + H2
CaCl2 + H2
Tests for hydrogen can be conducted in both reactions by holding a lit splint close to the top of the tube.